|
12/7/2023 0 Comments Draw the lewis structure for sf2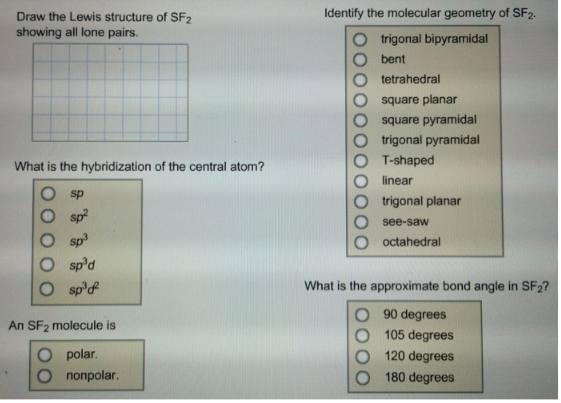
The structure of SF 4 can therefore be anticipated using the principles of VSEPR theory: it is a see-saw shape, with S at the center. Of sulfur's total of six valence electrons, two form a lone pair. 
Sulfur in SF 4 is in the formal +4 oxidation state. 3 Use of SF 4 for the synthesis of fluorocarbons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
